Ovulation Detection Using Ultrasound
October 21, 2022
Ultrasound-Assisted Rhinoplasty Procedure
December 5, 2022The intrauterine contraceptive device (IUCD), often referred to as the intrauterine device (IUD) and more frequently as the coil, is one of the most widely used methods of contraception in the world. It prevents pregnancies by thinning the endometrial lining, halting sperm motility, and preventing implantation.
The intrauterine device (IUD) is becoming more and more popular as a reversible method of birth control. When a patient has pelvic pain, unusual bleeding, or no retrieval strings, ultrasound is the imaging method of choice for determining the IUD’s position.
Eviction, displacement, embedment, and perforation are issues that might happen after implantation even for the most experienced clinicians. Transvaginal ultrasonography can make the operation simpler and minimize risk. According to studies, uterine perforation occurs around once every 1,000 insertions. (According to Teal SB, Sheeder J (2012), IUD use in adolescent mothers)
Therefore, in order to anticipate whether an intrauterine contraceptive device (IUD) will be retained successfully, an ultrasound examination is used to evaluate the success of IUD insertion immediately following delivery and to identify the ideal distance between the lower end of the IUD and the internal organs.
Which ultrasound is suitable for Monitoring IUD?
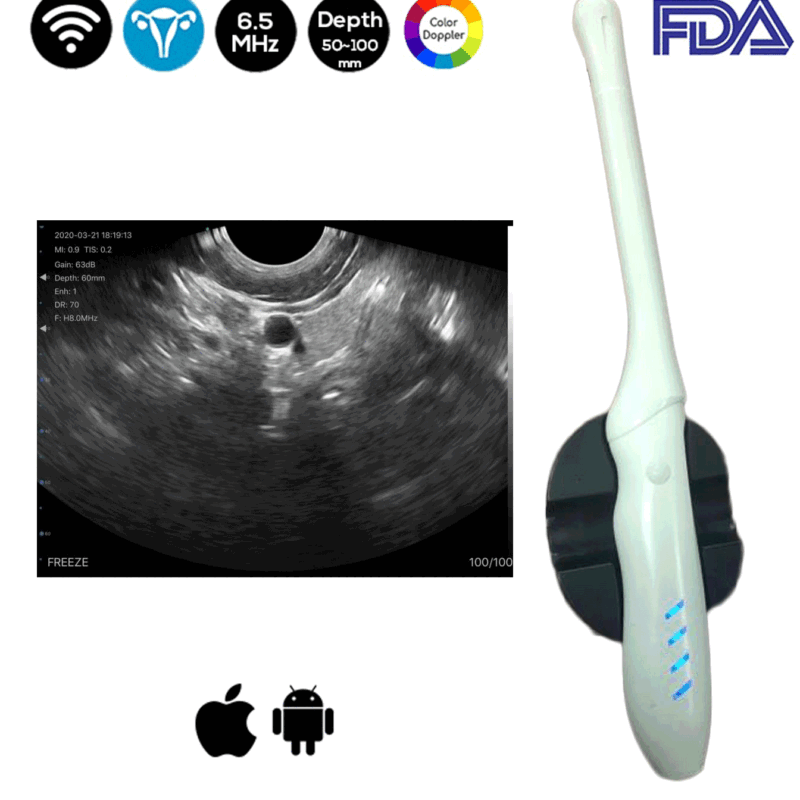
Convex and Transvaginal Color Doppler Double Head Wireless Ultrasound Scanner CTC-3.1 with 6.5Mhz is used by our OB-GYN to provide a scan at a depth of 50-100mm to the implantation of an intrauterine device (IUD).
The Wireless Ultrasound Scanner comes handy throughout the whole procedure. IUDs are placed in an outpatient setting using readily available kits and a sterile technique.
A sterile uterine sound is used to ensure a uterine depth of at least 6 cm. Image guidance is typically reserved for women with a history of difficult insertion, obesity that limits bimanual exam, or suspected uterine cavity distorted. A 6-week follow-up ultrasound pelvic exam is recommended to ensure visualization of the retrieval strings, which should protrude through the external cervical os by 2-3 cm.
Disclaimer: Although the information we provide is used by different doctors and medical staff to perform their procedures and clinical applications, the information contained in this article is for consideration only. SONOSIF is not responsible neither for the misuse of the device nor for the wrong or random generalizability of the device in all clinical applications or procedures mentioned in our articles. Users must have the proper training and skills to perform the procedure with each ultrasound scanner device.
The products mentioned in this article are only for sale to medical staff (doctors, nurses, certified practitioners, etc.) or to private users assisted by or under the supervision of a medical professional.